Farmaceutski otvorena ispitna platforma za nano-farmaceutske inovativne proizvode
Službena web-stranica: https://www.phoenix-oitb.eu/
Akronim projekta: PHOENIX
Trajanje projekta: 1. 3. 2021. – 28. 2. 2025.
Koordinator projekta: MyBiotech, Njemačka
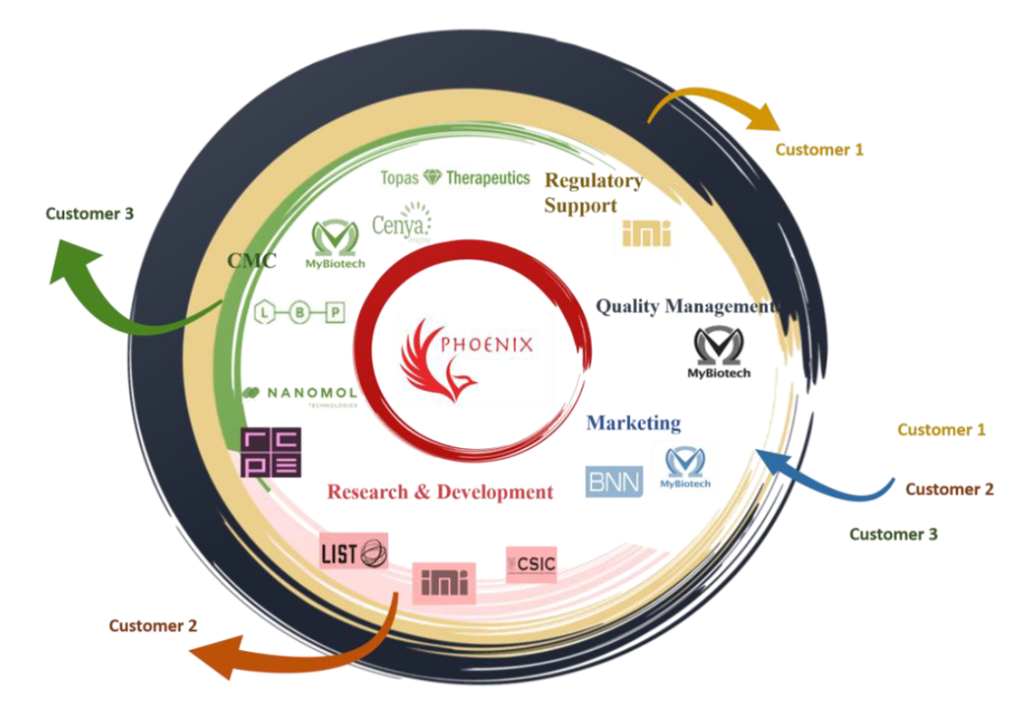
Naša uloga: Voditelj Radnog paketa 5 (WP5) – Regulatorna podrška
Financira: European Union Horizon 2020 Programme (H2020) under grant agreement nº 953110
Proračun: 11.118.925,26 EUR
Sažetak: Nanofarmaceutski proizvodi imaju potencijal za poticanje znanstvenog i tehnološkog razvoja zemalja članica EU, nudeći velike kliničke i socioekonomske koristi društvu u cjelini, industriji i ključnim dionicima te konačno pacijentima. Pristupačna i napredna ispitivanja, proizvodni pogoni i usluge za nove nano-lijekove glavni su preduvjeti za uspješnu provedbu ovih dostignuća kako bi se dodatno ojačali rast i inovacijski kapaciteti. Uspostavljanje trenutne dobre proizvodne prakse (GMP od eng. good manufacturing practice) u nano-farmaceutskoj proizvodnji ključni je korak za uspješan prijenos nano-lijekova iz laboratorija u industrijsku razinu, te kliničku praksu. Zbog nedostatka resursa za provedbu GMP proizvodnje, razvoj i proizvodnja inovativnih nano-lijekova i dalje predstavlja izazov glavnim igračima na tržištu nanomedicine u EU, novoosnovanim poduzećima te malim i srednjim poduzećima. Da bi se omogućila uspješna primjena nano-lijekova u području medicine, hitno je potrebno uspostaviti Otvorenu inovacijsku testnu platformu (OITB od eng. open innovation test bed) temeljenu na znanstvenim dostignućima i regulatornim pravilima. Cilj PHOENIX projekta je omogućiti neometani, pravodobni i cjenovno prihvatljivi transfer nano-lijekova iz laboratorija prema kliničkim ispitivanjima pružanjem napredne, pristupačne i lako dostupne PHOENIX platforme. PHOENIX platforma će ponuditi konsolidiranu mrežu laboratorija, proizvodnih pogona, tehnologija, usluga i stručnosti za sve aspekte prijenosa tehnologije, od karakterizacije, ispitivanja, verifikacije do konačnih proizvoda usklađenih s GMP proizvodnim i regulatornim smjernicama. PHOENIX platforma će razviti i uspostaviti nove pogone i nadograditi postojeće kako bi ih učinili dostupnima malim i srednjim poduzećima, novoosnovanim i istraživačkim laboratorijima za proširenje, proizvodnju GMP-a i ispitivanje nano-lijekova. Usluge i stručnost koje pruža OITB uključivat će proizvodnju i karakterizaciju pod GMP uvjetima, procjenu sigurnosti, usklađenost s propisima i jačanje komercijalizacije.
Projekt partneri:
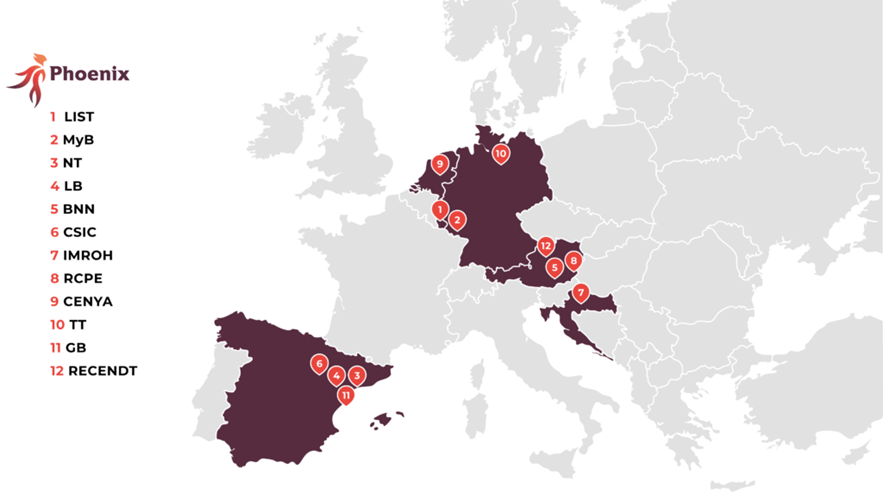
MyBiotech GmbH (Njemačka) – Koordinator
Nanomol Technologies SL (Španjolska)
LeanBio SL (Španjolska)
BioNanoNet Forschungsgesellscchaft mbH (Austrija)
Luxembourg Institute of Science and Technology (Luxembourg)
Agencia Estatal Consejo Superior Deinvestigaciones Científicas (Španjolska)
Institute for Medical Research and Occupational Health (Hrvatska)
Research Center Pharmaceutical Engineering GmbH (Austrija)
Cenya Imaging B.V. (Nizozemska)
Topas Therapeutics GmbH (Njemačka)
Grace Bio S.L. (Španjolska)
RECENDT (Austrija)