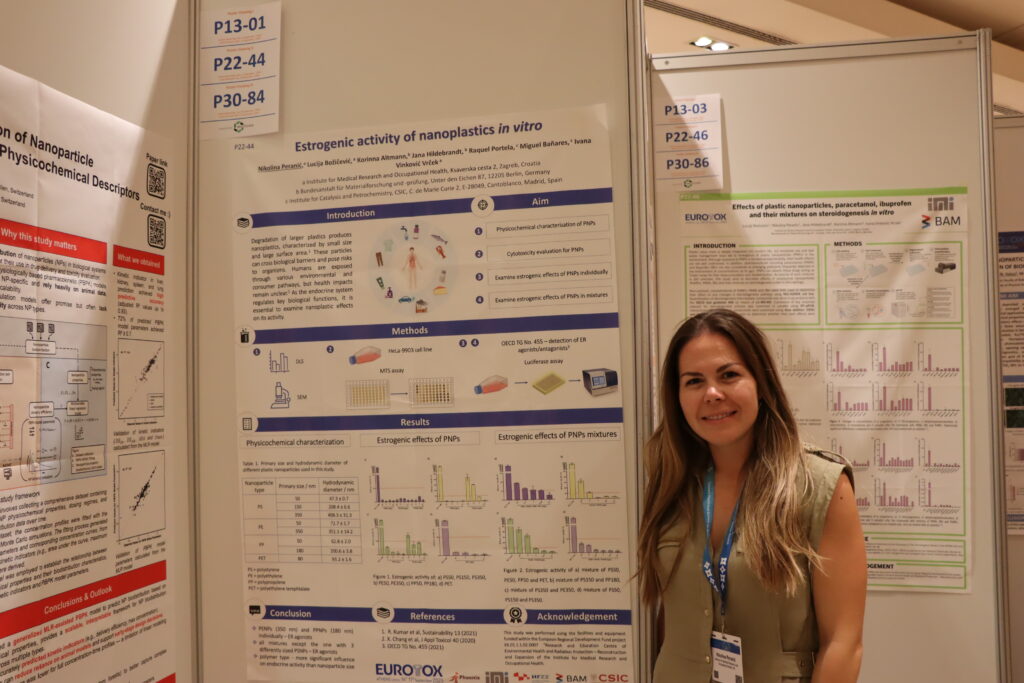
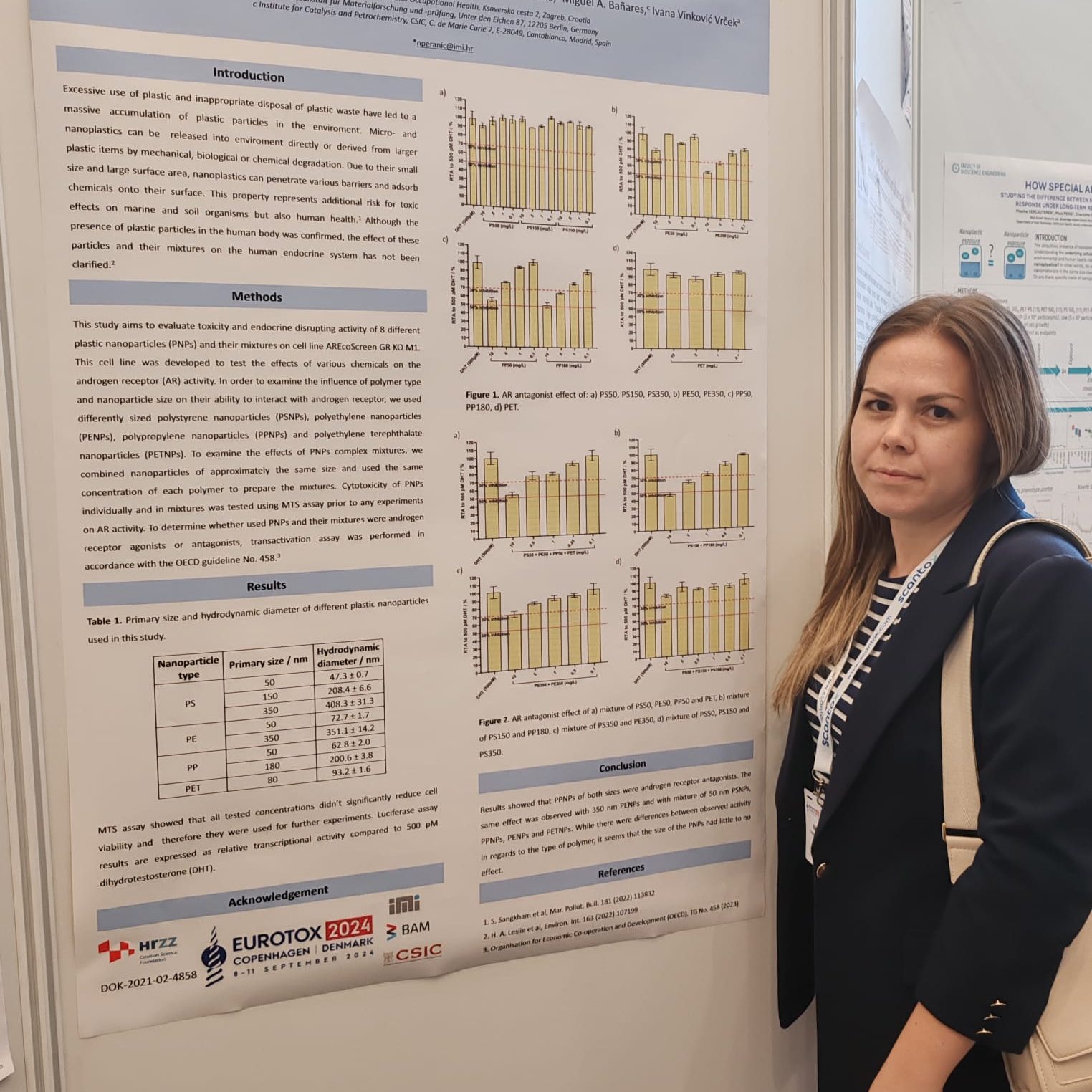
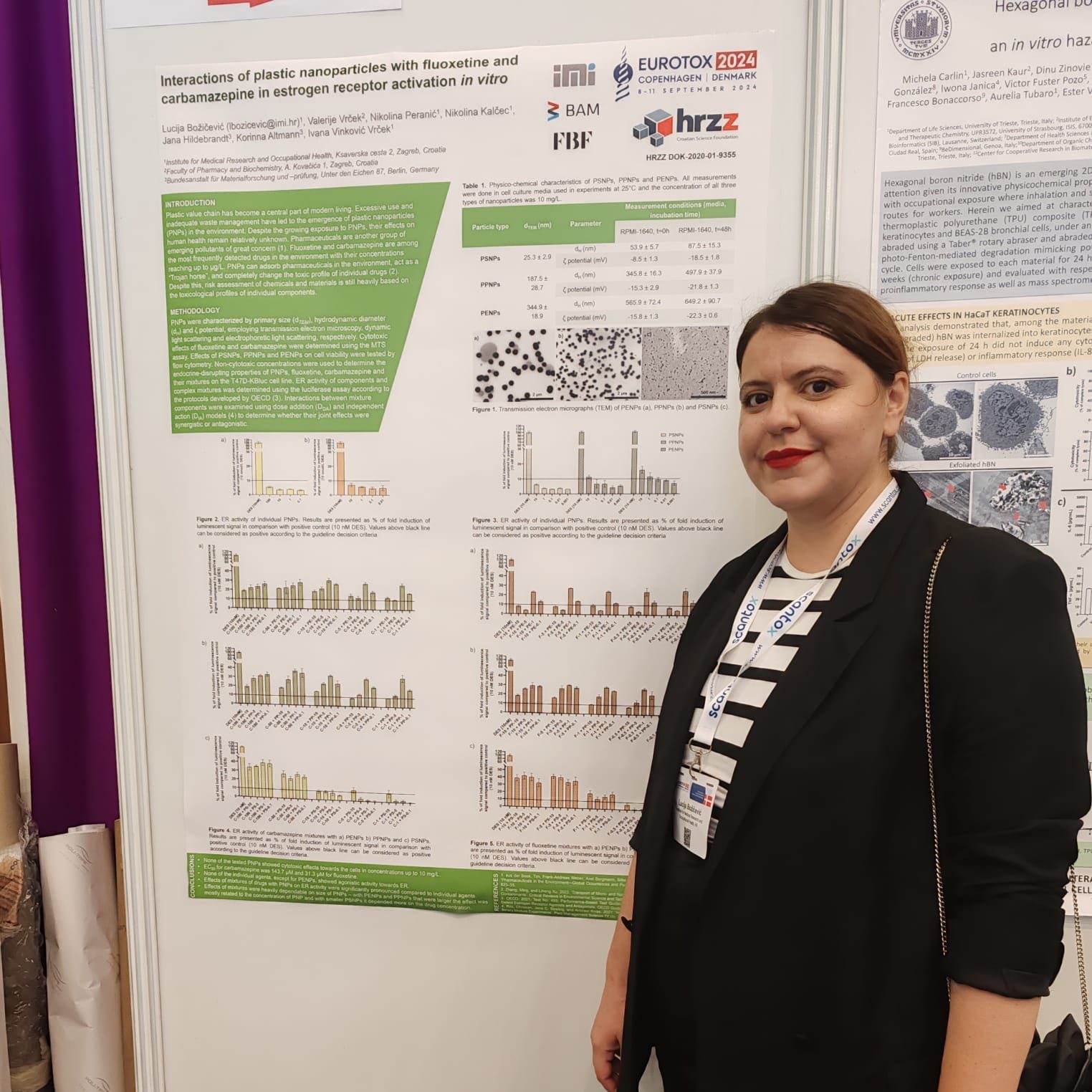
The Organisation for Economic Co-operation and Development (OECD) has published a new edition of its guidelines titled “Guidance on Sample Preparation and Dosimetry for Manufactured Nanomaterials (2025 Edition).” The document introduces updated standards for sample preparation and dosimetry in nanomaterial safety testing, reflecting the latest scientific developments in the field.
Titular professor Ivana Vinković Vrček, PhD, from the Institute for Medical Research and Occupational Health (IMI), participated in the development of this international standard. Alongside Tommaso Serchi (LIST, Luxembourg), she led the drafting of Chapter VII, which focuses on toxicological testing, including both in vitro and in vivo methods. The participation of IMI scientists in these working groups continuously contributes to the harmonization of global and national protocols for the safe application of new technologies.
The complete document is available via the OECD iLibrary